Key Features:
- It manages the complete flow from initiation of CCP to closure after effective review.
- It is linked to other QMS modules like DMS(DocsExecutive) and eTraining.
- Architecture allows application access using web technology.
- Electronic Signatures and audit trails as per 21 CFR Part 11 guidelines of USFDA.
- Default impacted departments can be loaded as per product type or category, so user need not remember everytime when adding impacted departments.
- Each HOD from impacted departments can add action points as per CCP requirements with target dates.
- Auto task reminders through emails and notifications based on target dates given for action points and other activities like review/approve/impact analysis.
- CCP can be monitored OR tracked by Site QA using different tracking options.
- MIS – Department and Locationwise Trend analysis report
CCP Flowchart:
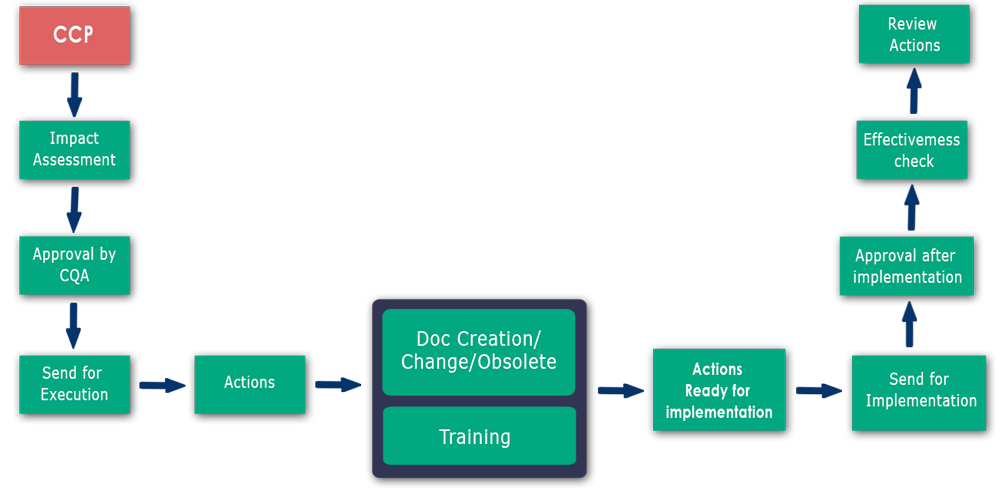
-->
-->
-->