Loading...
Loading...
Comprehensive regulatory and quality management system software designed to accelerate your pharmaceutical business success with streamlined compliance and automation.



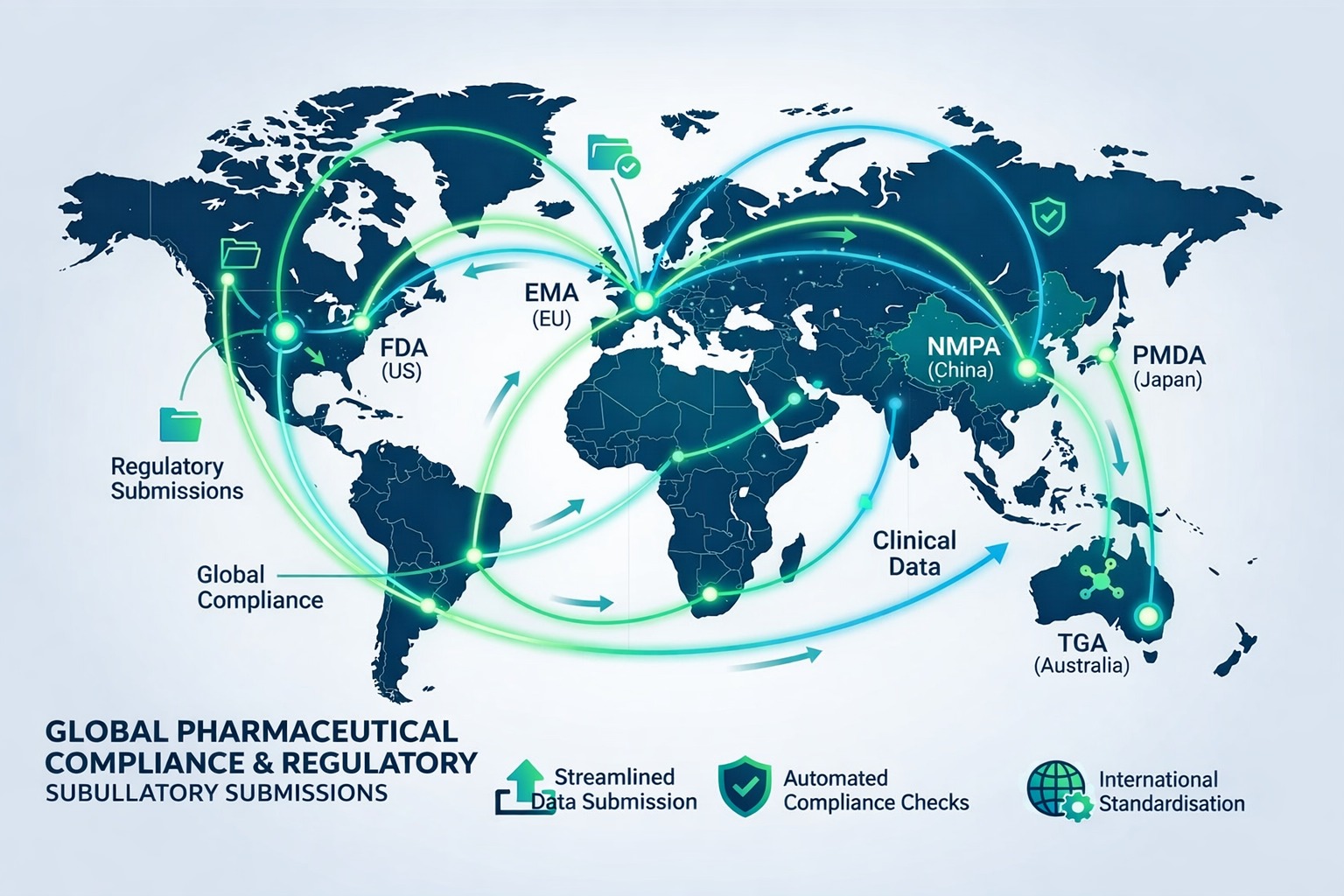
Experience Educe Solutions' deep expertise in electronic dossier management and global regulatory compliance. Our user-friendly software, responsive technical support, and commitment to innovation are recognized and recommended by leading pharmaceutical organizations worldwide.
Our commitment to your success, validated by experience and performance.
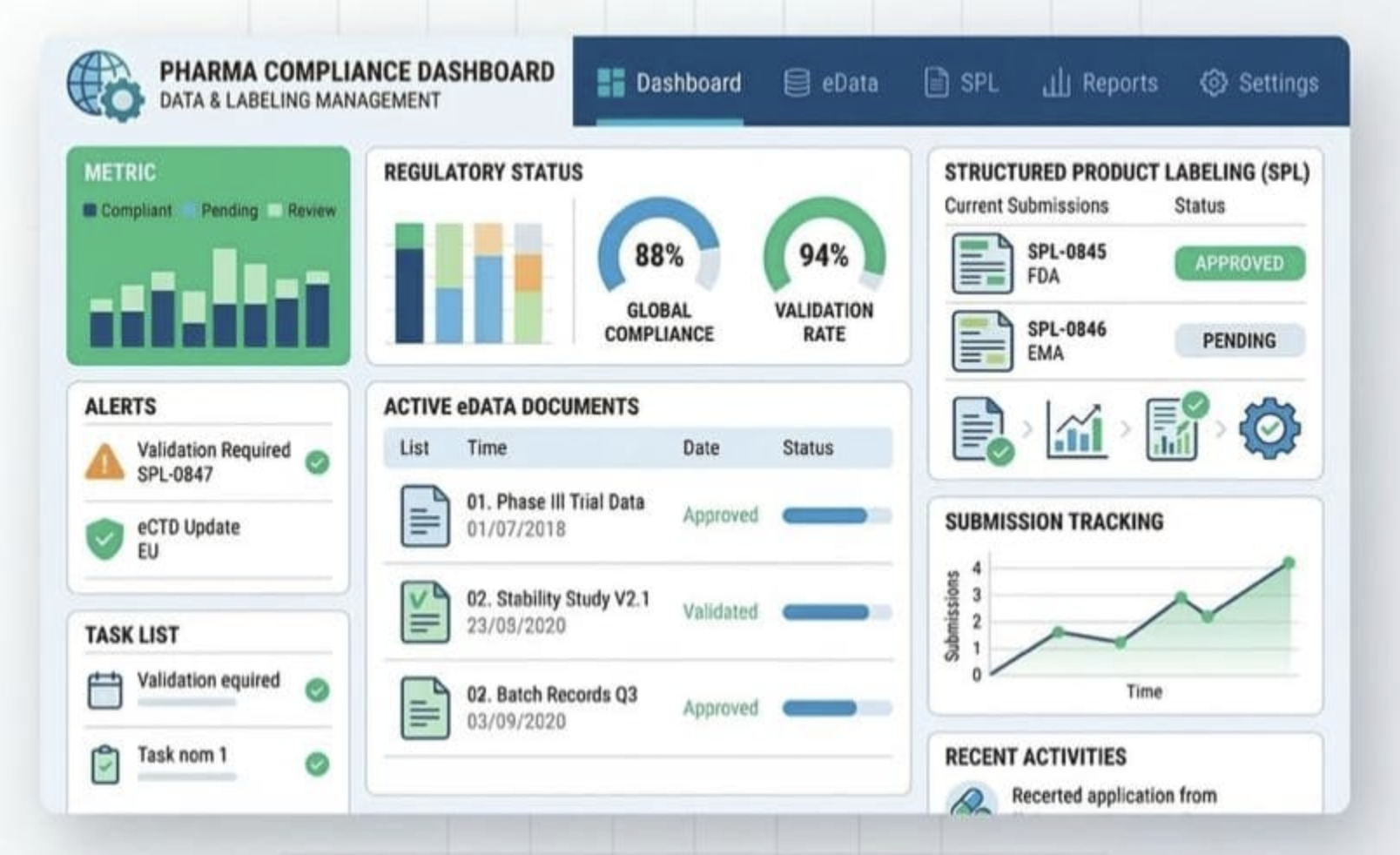
Dossier-Mgmt© empowers pharma teams to automate dossier creation, ensure compliance, and speed up global submissions, reducing manual effort and approval delays.

Our commitment to your success, validated by experience and performance.
"I've been using Educe Solutions for over a decade. Their reliable, easy-to-use eCTD and SPL software simplifies compliant submissions. The team is very responsive and truly understands regulatory needs and they're a trusted long-term partner. I can confidently recommend Educe Solutions to any organization seeking excellence in electronic dossier management."
"For educe, I would like to say that we have always received the required support from the team. I would like to believe that we were one of the few initial customers and now happy to have this long term association with Educe. The team is technically strong, very cooperative and extends support whenever needed."
"We have been using Educe Solutions’ eCTD software for our regulatory submissions for the last 5 years, and it has significantly streamlined our processes. The user-friendly interface, robust validation checks, quick support from support team, and seamless publishing capabilities have ensured full compliance with global health authority requirements. Educe Solutions’ software also provides a reliance checklist to fulfil the requirements of the global medicinal authority. We have been served by the Educe team for the submission in various regulated region like USA, Europe, Australia along with the semi-regulated Gulf countries. Their team is highly responsive and supportive, making implementation and ongoing use smooth and efficient. Educe Solutions is not just a software provider but a trusted partner in regulatory success. We would recommend to have this software for any organization for error-free smooth regulatory submission."
"Educe Solutions = Ease of use, user-friendly interface, immediate and dedicated customer service & support function, rapid troubleshooting, immediate solutions, our preferred regulatory software for global submissions."
Educe's software is built around industry standards and regulations. For example:
Our pharma software is designed to streamline your regulatory workflow, enhance compliance, and automate quality processes so your team can focus on what truly matters.
Let's talk! We'll reply within a day. Get Free Quote →
Understanding your regulatory and quality needs
Customized software and process planning
Seamless deployment with dedicated support
Ongoing optimization for compliance and business goals